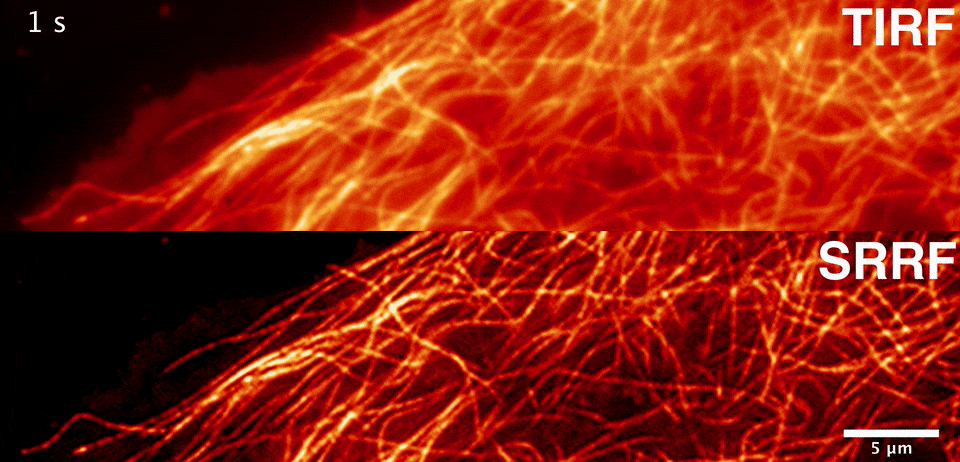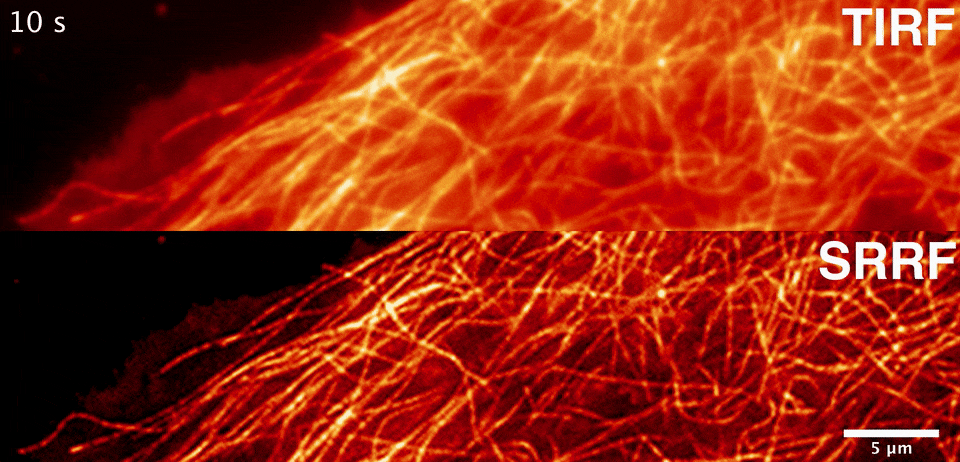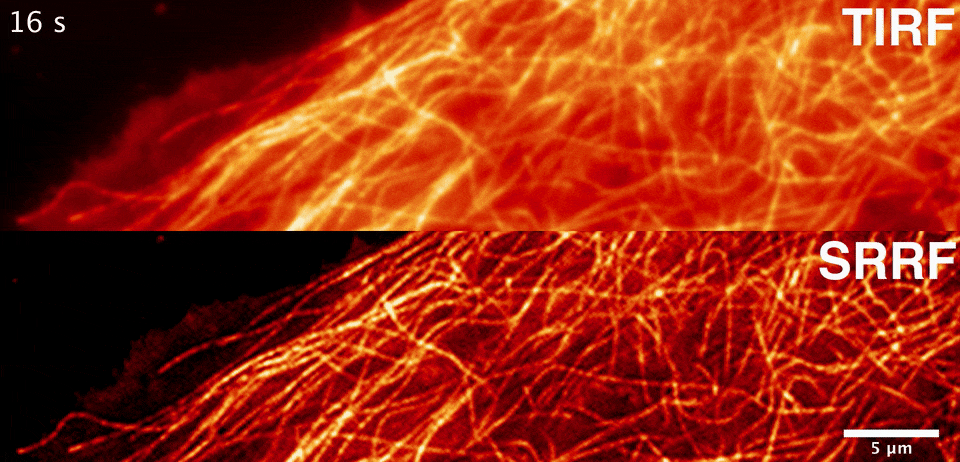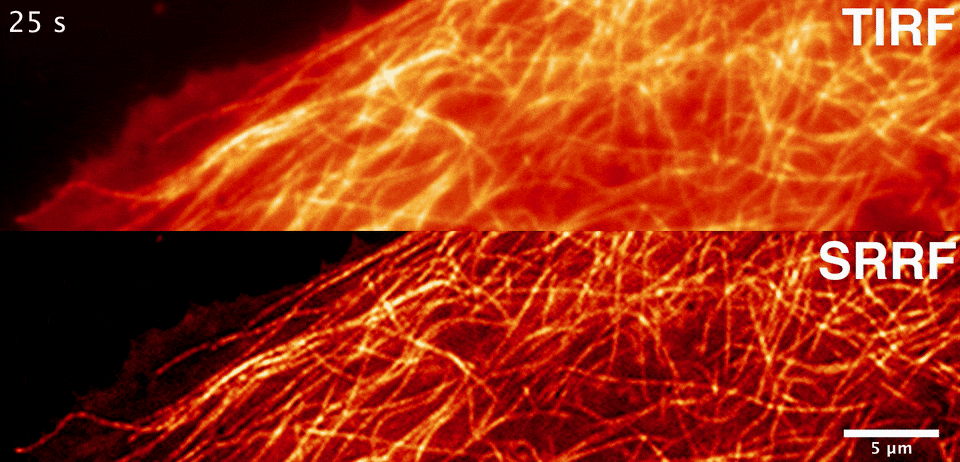
Sublimation energy of Na 26 kcal//gatom, di asked in Chemistry by siddhiChawla ( 59. What would happen to the lattice energy between ions if the distance increases to 2 angstroms? A. Calculate the lattice energy of NaCI crystal from the following data by the use of Born-Haber cycle. Two atoms with charges of +1 and -1 are separated by a distance of 0.5 angstroms. Consider the binding energy per nucleon for Would you expect the fusion reactions leading from 16-O to 56-Fe to be exoergic (giving off energy), endoergic (absorbing energy), or neither? 2. Consider the binding energy per nucleon for 16-O and 56-Fe. The energy varies during the catalyzed reaction Thanks in advance, I appreciate it :)ġ. What happens to the energy of the transition state when a catalyst is used? a. In this reaction, a simple ionic compound (Sodium chloride salt) is separated into its ions in gaseous forms (Sodium and Chlorine), but to proceed with or complete this reaction, some amount of energy is required, which is known as lattice energy of the ionic compound. Select one a CsBr s b NaCl s c SrO s d CaO s e KBr s The correct answer is. Rank the following ionic compounds by lattice energy. If the space lattice is SC, the lattice constant is given by the formula a. Which of these kinds of energy is a fundamental requisite for life on Earth? A. What is a complete ionic equation for the reaction between Pb(NO3)2 and NaI? they are both key components of plant cells. The calculation of lattice energy can be done by using Hess’s law (for this case it is called Born Haber cycle). Here we can see that the lattice energy of MgO is much greater than the lattice energy of NaCl. they both use energy transport chains.*** d. Thus +2 or -2 ions will release more lattice energy than the +1 or -1 ions. they are both key components of solar panels. they both produce chemical and electrical energy. ATP and photovoltaic cells are similar because a. Solution: The lattice energy of NaCl represents the energy required to break solid NaCl into its components gaseous ions.

Biology - Photosynthesis: Energy Conversion Quickġ. Calculate the lattice energy of NaCl using the Born-Haber cycle.How many electrons are in each energy level?(1 point) 1.two in the first energy level, six in the second energy level 2.eight in the first energy level, zero in the CsIĪ model of an atom shows eight electrons in rings that represent different energy levels. Which of the following ionic compounds has the highest melting point? Help: Lattice energy Select one: a. (iii) The lattice energy affects the solubility of the ionic compounds.Calculate the lattice energy of sodium oxide (Na2O) from the following data: Ionization energy of Na(g): 495 kJ/mol Electron affinity of O2 for 2e: 603 kJ/mol Energy to vaporize Na(s): 109 kJ/mol O2(g) bond energy: 499 kJ/mol Energy change for the reaction (ii) The lattice energy is greater for small, highly charged ions. With the NaCl Lattice Magnetic Model, take salt crystal lessons from 2-D to 3-D using the sodium chloride ion model Students discover the cubic nature of. (i) The greater the lattice enthalpy, the more the stability of ionic compounds. The important consequence of the lattice energies are: $\therefore $The order of lattice energy is $NaF > NaCl > KCl > RbCl$. The given ionic solids are $NaCl,$ion so $NaCl$has greater lattice energy than $KCl$ and $RbCl$. Hence, more will be the value of lattice energy. smaller the size of ions, lesser is the internuclear distance and greater will be the interionic attraction. larger the magnitude of charge on ions, greater will be the attractive forces, higher is the value of lattice energy and inversely proportional to size of an ion i.e. APF for a simple cubic structure 0.52 atoms unit cell RO.5a close-packed directions contains 8 x 1/8 1 atom/ unit cell Adapted from Fig. Lattice energy is directly proportional to charge on ion i.e. Lattice points Lattice points are theoretical points arranged periodically in 3-D space. Lattice energy depends upon charge and size of cation. Hint: Lattice energy: The amount of energy released when $1$mole of an ionic solid is formed from its gaseous ions.
